Our Focus
The common thread among diseases we are targeting is lysosomal dysfunction caused by heterozygous loss-of-function mutations in lysosomal genes. This haploinsufficiency adversely affects the autophagy-lysosomal pathway (ALP), disturbances of which are known to be associated with neurodegeneration¹,².
Over the last decade or so, there has been an emergence of evidence suggesting that heterozygosity in certain lysosomal genes is associated with Parkinson’s disease (PD)³, Frontotemporal Dementia⁴, and most recently AD⁵. We believe that up to 30% of all patients suffering from various neurodegenerative diseases harbor pathogenic mutations in lysosomal genes.
Our future therapies have the potential to restore the normal lysosomal gene function and prevent disease progression. In addition to treating neurodegenerative diseases associated with lysosomal dysfunction, the therapies we’re developing may have the potential to treat the CNS manifestations of lysosomal storage diseases.

Alzheimer’s Disease
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that kills more than breast cancer and prostate cancer combined. It is the most common cause of dementia, impacting nearly 7 million Americans (1 in 9 people aged 65 or older has AD) and 55 million people worldwide, and is expected to double by 2050. In addition to 7 million AD patients in the US, there are about 5 million Americans diagnosed with Mild Cognitive Impairment (MCI) due to AD.
AD and MCI due to AD represent a major unmet need and, in 2024, will cost the payers approximately $360B. Not surprisingly, AD has been the major focus of the pharmaceutical industry for the past few decades, but despite billions of development dollars spent, over 200 clinical trials have failed, and currently, there is no cure. Many efforts focused on developing drugs that remove established amyloid-β (Aβ) plaques and tau tangles that accumulate in the brains of AD patients. While the accumulation of Aβ plaques and tau tangles is the hallmark sign of the disease, the underlying cause of AD in most people is poorly understood, and it appears to be driven by a combination of genetic factors. The APOE ε4 allele is a known genetic risk factor, especially for late-onset Alzheimer's. Early-onset Alzheimer's, which occurs before age 65, is often linked to mutations in APP, PSEN1, and PSEN2 genes.
Our discoveries suggest that heterozygous mutations in lysosomal genes are associated with AD⁶,⁷, a notion recently validated by an independent study⁵. Our strategy is to target the upstream effector pathways instead of downstream markers in order to prevent disease progression.
Parkinson's Disease
Parkinson's disease (PD) is a chronic and progressive neurodegenerative disorder that primarily affects movement. It occurs due to the gradual loss of dopamine-producing neurons in a region of the brain called the substantia nigra, which is part of the basal ganglia. Parkinson’s disease affects about 1 million people in the US and is the second most common neurodegenerative disorder after Alzheimer’s.
The exact cause of Parkinson's disease is unknown, but it is believed to be due to a combination of genetic and environmental factors. Environmental factors, such as exposure to pesticides and heavy metals, have been associated with an increased risk of developing the disease. Genetic factors include mutations in LRRK2, PINK1, PARKIN, SNCA, and the lysosomal gene GBA³. In fact, heterozygosity of GBA is now recognized as one of the highest risk factors for PD, and it is estimated that about 10% of individuals with PD carry a GBA mutation, although the prevalence may be higher in certain populations.
Our genetic studies have identified other lysosomal genes that are enriched in PD that may present therapeutic opportunities in addition to those targeting GBA. Overall, this shows that PD is not a homogenous disease, and there are distinct patient subpopulations, each with a unique disease origin.

Frontotemporal Dementia
Frontotemporal dementia (FTD) is a group of neurodegenerative disorders characterized by progressive damage to the frontal and/or temporal lobes of the brain, which control decision-making, behavior, emotion, and language. As a result, FTD typically leads to significant changes in personality, behavior, and language skills, but can also affect movement.
FTD is less common than other types of dementia, such as Alzheimer's disease, and it affects approximately 50,000 to 60,000 people in the United States, although the actual prevalence is not known. FTD typically has an earlier onset than Alzheimer's, often affecting people as early as 45 years of age. Because of its earlier onset and different symptom profile, FTD is often underdiagnosed or misdiagnosed.
FTD is often linked to abnormal protein accumulations in the brain, particularly of tau and TDP-43 proteins. While the exact cause of FTD is not fully understood, research suggests that the most significant risk factor is family history, especially with known genetic mutations in C9orf72, MAPT or heterozygosity in the lysosomal gene GRN⁴.
Lysosomal Storage Diseases
Lysosomal storage diseases or disorders (LSDs) are a group of over 70 rare genetic diseases that are associated with significant morbidity and mortality. Although individually classified as rare diseases, collectively, LSDs affect approximately 1 in 5,000 live births. Most are inherited in an autosomal recessive manner⁸. LSDs are characterized by a specific lysosomal enzyme deficiency or dysfunction leading to abnormal accumulation of substrates causing cellular, tissue, and organ damage.
Though many organs may be affected, the majority of LSDs involve severe neurodegeneration, including at an early age⁹. Regardless of the age of onset, LSDs are relentlessly progressive and represent a significant burden to individuals, their families, and the healthcare systems, as only a handful of LSDs have available therapies and no cures. Therapies being developed by JayaBio have the potential to be used for the CNS manifestations of LSDs that overlap with our gene targets.
-
1. The Autophagy-Lysosomal Pathway in Neurodegeneration: A TFEB Perspective - Martini-Stoica H, et al. (2016) Trends Neurosci 39(4):221-234
2. Biological Functions of Autophagy Genes: A Disease Perspective - Levine B, Kroemer G, (2019) Cell 176(1-2):11-42
3. Heterozygosity of GBA is associated with Parkinson’s Disease - Sidransky E, et al., (2009) NEJM, 351:1972
4. Heterozygosity of PGRN is associated with Frontotemporal Dementia - Baker M, et al., (2006) Nature, 442:916
5. Heterozygosity of NPC1 is associated with AD - Lopergolo D, et al., (2024) J Med Genet, Epub ahead of print.
6. Haploinsufficiency of lysosomal enzyme genes and Alzheimer’s disease – Sands MS, et al., 20th Annual WORLD Symposium, February 4-9, San Diego, CA.
7. Haploinsufficiency of lysosomal enzyme genes and Alzheimer disease – Sands MS, et al., 45th Annual Meeting of the Society for Inherited Metabolic Disorders, April 14-17, Charlotte, NC.
8. Recessive means that two copies of the mutated gene (one from each parent) are required to cause the disorder.
9. Lysosomal storage diseases - Frances M. Platt, et al., (2018) Nature Reviews Disease Primers, V4:2
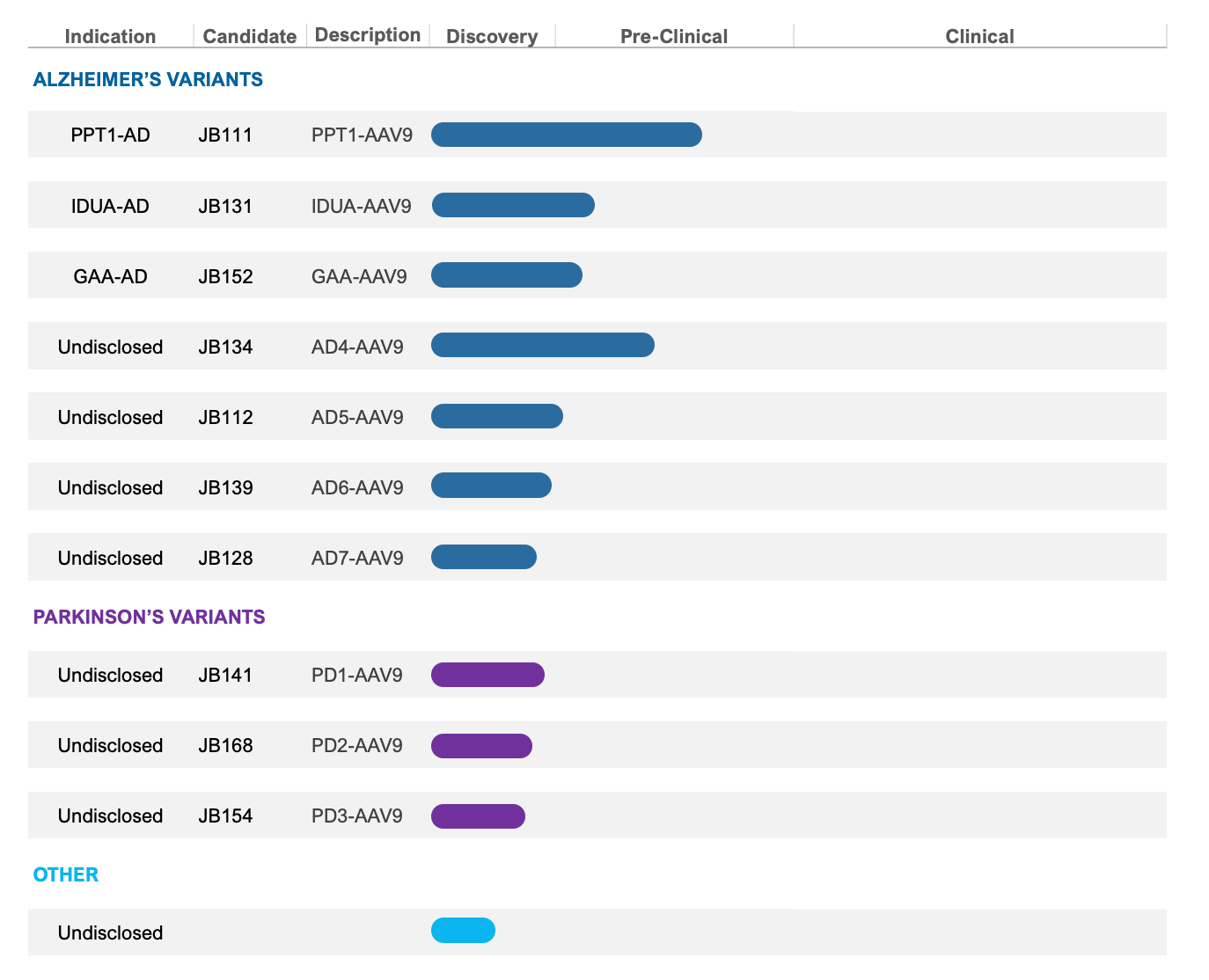
Pipeline